Smart ring market leader Oura is trying to convince U.S. lawmakers to loosen some health monitoring regulations for wearable devices, which if successful will benefit Apple too. Users will just need to be careful about what they do with the data.
Products going on sale must meet regulatory requirements, and it's something that Apple is very familiar with for its product line. However, if work by one wearables company results in changes in the United States, that could directly benefit Apple's own products, too.
Smart ring maker Oura stepped up its work to leave a good impression on the U.S. government in 2025, a Politico report details. As well as handing out smart rings to Olympic athletes, it has increased its lobbying budget.
While it spent just $40,000 in 2024, the figure exceeded $1 million in 2025.
The effort hasn't gone unnoticed, with Oura popular in Washington and counting the Department of Defense as its largest customer. There have also been efforts by lawmakers to try and protect Oura from rivals in India and China, even though it's a Finnish company.
Its latest focus is on U.S. policy decisions, with the Food and Drug Administration's medical-grade classifications in its sights.
Currently, the FDA regulates wearable devices as being either a general wellness product with little oversight or a medical device. Hardware designated as a medical device would require extra reviews for safety and effectiveness, which eats up time and resources.
Oura chief executive Tom Hale wrote an op-ed in December calling for the FDA and Congress to create an exemption for wearable devices, to help them avoid the dreaded medical grade classification. He insists there should be a third category available for "digital health screeners," intended for devices to warn of health issues without performing an actual diagnosis.
The op-ed seemingly helped, as the FDA changed its rules so that wearables could warn users to seek a more thorough medical evaluation from a physician. It also opened the door to devices measuring blood pressure and blood sugar without necessarily needing the medical device approval beforehand.
Oura isn't finished yet, as it has called for lawmakers in Congress to make the changes into law, and for further clarity on what features should be allowed without needing the extended approvals.
It may get its way here too, as Rep. Troy Balderson (R-Ohio), chair of the House digital health caucus, is working on legislation in this area. Balderson said that the legislation is to create an environment for wearables to expand and be more innovative.
Risky business
The discussion of legislatively freeing up companies to create new features without needing to undergo a rigorous review process is generally framed as a pro-innovation initiative. Even so, such a policy introduces risk.
The point of the FDA reviews is to ensure that, if a device claims to measure a health-related statistic, it does so reliably. When it comes to monitoring vital data points about the body, such as blood glucose levels, an incorrect reading could result in a user responding in a way that actually endangers their life, or failing to respond when they really need to.
In February 2024, the FDA went on record to say that non-invasive glucose monitoring had not yet been approved due to fears of inaccuracy. It warned that patients shouldn't buy devices specifically for this purpose, and to continue using direct blood monitoring.
Apple has long been said to be testing glucose monitoring features, with a view to adding it to the Apple Watch in the future.
So far, it has only really manifested on the Apple Watch in the form of a Dexcom continuous glucose monitor, not an Apple-produced technology.
The FDA already has to deal with problematic tech when it comes to glucose monitoring. In December, the FDA warned consumers against the use of some sensing devices, which were linked to at least seven deaths and over 700 serious injuries.
The faulty devices fed incorrect readings to users, who then treated their condition based on that wrong data. In these reported cases, it led to users inadvertently harming themselves, sometimes fatally.
Even so, getting some data to work with is better than not having data at all. As we have seen many times before, the Apple Watch hasn't actually made a diagnosis, but it has nudged users to seek out professional help with its various health-related warnings.
All of this without being strictly a medical-grade device. It does, however, require that users be smart about what data is shown to them.
Oura isn't the only company lobbying for changes, but not everyone is following the same line. Rival wearables maker Whoop was happy about the FDA making physiological metric rule changes for wearables, but didn't endorse creating a whole new "digital health screener" category.
Alex Vannoni, the VP of health care at Whoop, urged "caution" when considering the creation of an unregulated and undefined category for this purpose.
An Apple (device) a day...
Oura's attempts to convince the U.S. government to be more hands-off with wearable devices has the potential to affect more than just Oura or smart rings in general. It could apply to many other wearable devices.
For Apple, this would obviously cover the Apple Watch lineup, as well as heart rate sensors such as in the AirPods Pro 3 and the Beats Powerbeats Pro 2.
There's also the repeatedly rumored Apple Ring, a smart ring that has repeatedly surfaced in rumors but has still failed to make it to market. Depending on the rumor you read, it could provide anything from limited health tracking to gesture recognition.
At the moment, the Apple Watch would be the main beneficiary from FDA or Congress-based classification changes.
While it hasn't been needed for every feature, such as the introduction of blood oxygen sensing in the Apple Watch Series 6, other features have had to jump through more regulatory hoops.
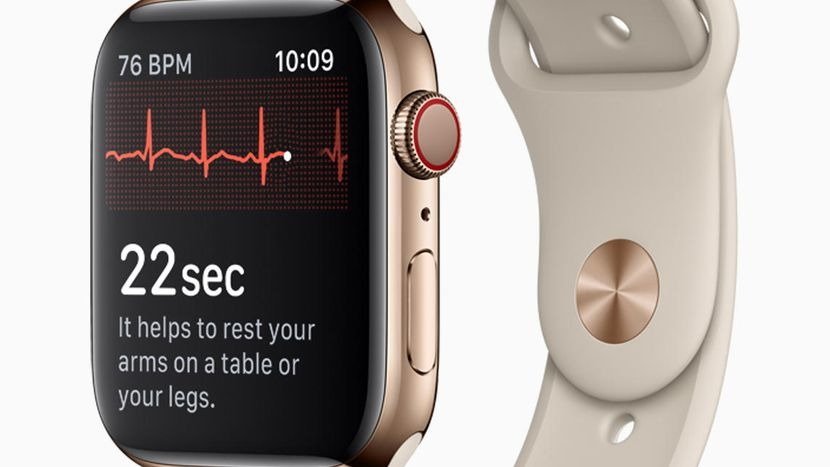
Its inclusion of an ECG in the Apple Watch Series 4 was groundbreaking, but FDA clearance arrived less than 24 hours before its launch. It was a similar story for the Atrial Fibrillation feature, with it approved hours before Apple's WWDC 2022 keynote.
Securing features like these and hypertension notifications requires a lot more documentation, research, and evidence gathering to submit to the FDA for its review. Even then, there's no guarantee that the FDA will provide a fast-enough response, nor will it necessarily grant that approval.
A reclassification would free Apple up from these constraints, and to more easily roll out the features to consumers.
If it had already been adopted, Apple would've certainly brought features like non-invasive glucose monitoring to the Apple Watch by now, instead of continuing to develop it over the long term prior to release.
Care is still needed
Oura may get its way and help Apple and others develop health-based features and bring them to market faster. But doing so has the potential to risk the lives of users if it is not implemented with the right safeguards in place.
While companies in the space may be emboldened to bring out new features thanks to a reduced level of regulatory oversight, it's a big risk to take. Though companies may foresee a rise in potential lawsuits from the data those features bring out, users could end up paying the ultimate cost if they're not careful.